Overview of the genus Gyrodactylus
Gyrodactylus is a hyperdiverse monogenean genus with ca. 400 described species from a potential total of 25,000 host species (see Bakke et al. 2002). They are ubiquitous teleost ectoparasites that have been extensively studied since the late 19th Century, largely due their unique reproductive adaptations.
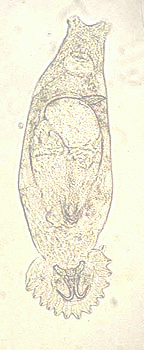
Gyrodactylids are the only parasitic worms that reproduce in situ on their host and lack a specific transmission stage, therefore they bridge the classical divide between micro-(bacteria, viruses etc) and macroparasites (worms etc). These viviparous parasites already contain a fully grown embryo (the F1) in utero when born and as the parent ages, development of a third generation embryo (the F2) is visible within the F1 (see Fig. 1). This “Russian-Doll” mode of reproduction, termed hyperviviparity, combined with a rapid generation time (<24h at 25oC for the 1st born daughter in some species), can result in exponential parasite population growth on a single host. A further complication is that these parasites can potentially employ different reproductive modes during their life cycle; asexual, parthenogenetic and sexual reproduction reviewed in Cable & Harris, 2002).
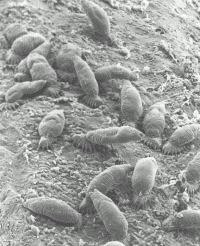
Newborn parasites attach to their host alongside their mothers using their posterior attachment organ, the opisthaptor (Fig. 2). They feed on mucus and epithelial cells, and can move freely on the host in almost a “caterpillar-like” fashion by alternatively attaching to the host’s epidermis with their posterior opisthaptor and anterior attachment glands. Many gyrodactylids exhibit distinct site preferences on the host, but may display characteristic migrations on the host during their life cycle.
Gyrodactylids are small (<1mm in length; Fig. 3A) and low numbers may appear to have little effect on their hosts, but initial appearances are misleading! Even low infection levels can cause host mortality and higher parasite burdens can cause secondary pathology, such as fin clamping (Fig. 3B), which presumably inflicts severe fitness costs to the host. The actual cause of host death is unknown, but may be induced by secondary infections and/or osmotic problems associated with the puncture wounds inflicted by gyrodactylids during attachment and feeding. Most host populations show evidence of an immune response against gyrodactylids (reviewed by Buchmann) with considerable within and between population heterogeneity in resistance.

With increasing aquacultural practices, the threat posed by gyrodactylids is ever more apparent, particularly since the havoc created by the G. salaris epidemics. This one species alone has cost the Norwegian Salmon Industry >500 million € in the first 25 years since its introduction from the Baltic in the 1970s. Apart from the financial costs to fish farming, this parasite endangers wild salmon stocks and is a significant conservation threat. With its high fecundity and transmission potential, G. salaris can rapidly colonize an entire river system (Bakke et al. 1992; Johnsen et al. 1999) and even re-colonize waterways after treatment with the rotenone (Johnsen et al. 1999). Rotenone poisoning of entire river systems was used, largely unsuccessfully, for many years, and now control measures involving heavy metal (aluminium) dosing are being trailed in Norway. This year (2005), millions of Euros have been spent already on G. salaris containment, but it remains to be seen whether aluminium dosing is an effective control measure in the field. In the current global economy, the risk of further introductions of gyrodactylid monogeneans is an indisputable threat. The biology of these pathogens predilects them for introduction (Kennedy 1994) and numerous species have been introduced to Europe. Unfortunately, the impact of such introductions to the ecosystem is currently unpredictable, and factors influencing the pathogenic-potential of Gyrodactylus spp. remain largely unknown.

In addition to the G. salaris problem, gyrodactylids impose major costs on the aquarium trade (importation of ornamental fish alone being worth 90 million €/pa to the EU economy, with total industry turnover of >2 billion €/pa) and cause significant disease in a range of cultured fish, including cod (e.g. Bauer et al. 1973; Appleby 1994; Mo & Lile 1998). Increased drug resistance has also been reported in gyrodactylids infecting farmed fish (Goven & Amend 1982).
The value of gyrodactylids in epidemiological models has been recognised for many years (e.g. Scott & Anderson 1984), and a number of statistical and computer models have been designed to analyse the risk of infections spreading to new rivers (Paisley et al. 1999; Hřgĺsen & Brun 2003) and study the biology of parasite populations (e.g. Harris et al. 1994). Age-structured population models have been used to assess the impact of disease-induced mortality at different stages of the life-cycle of salmonids (Clers 1993). Risk assessments have also been conducted to assess the risk posed to UK fisheries by gyrodactylosis (Peeler et al. 2004), but the value of such risk assessments hinges on correct identification of the parasite species.
Traditionally, gyrodactylid taxonomy was based on morphological characters alone. Malmberg (1970) formally described six subgenera (Gyrodactylus, Mesonephrotus, Paranephrotus, Metanephrotus, Neonephrotus and Limnonephrotus) on the basis of excretory system type and host group/habitat. He subsequently (1970, 1993) established species groups within each subgenus based on marginal hook morphology. However, gyrodactylids are frustratingly similar in appearance and difficulties in species identification are believed to have considerable hampered ecological studies. More recently, molecular studies (e.g. works by Cunningham, Hansen, Cable, Lumme and many others) have provided an independent assessment of this morphological work, and although Malmberg’s (1970) species groups have no formal taxonomic status, they have served as a practical means of handling the complexity of this genus for over three decades. Malmberg’s (1970) contribution to gyrodactylid biology in establishing a foundation for all other phylogenetic, taxonomic and ecological studies cannot be over emphasised.
Despite their morphological conservatism, it appears that gyrodactylids display a range of different behavioural traits associated with the diverse ecology of their teleost hosts (e.g. Cable et al. 2002; Olstad et al. submitted). With >400 described Gyrodactylus species, the potential for ecological and evolutionary studies is enormous. Apart from G. salaris, most work to date has focussed on stickleback and guppy gyrodactylid models. We feel the practical advantages of working with these model systems for studying host-parasite interactions are unsurpassed. Both hosts and parasites are easy and cheap to maintain in the laboratory; at 25oC, these guppy gyrodactylids have a generation time of >24h; and as they are skin ectoparasites the entire trajectory of infection can be monitored on a single anaesthetised host. With increased knowledge accumulating from both laboratory and field studies, we can begin to take a holistic approach to gyrodactylid systematics by analysing morphological, molecular, ecological and biogeographical trends.
|

